Role of catheter interventions in Congenital Heart Disease

16 Jan, 2020
Catheter intervention has a very big role to play in the diagnosis and treatment of congenital heart disease (CHD). Without catheter interventions, management of CHD is nearly impossible. Let us discuss about role of different catheter interventions in the management of CHD
Diagnostic catheterization
As non invasive techniques like echocardiography and CT angiography are becoming more and more advanced and user friendly, role of diagnostic catheterization is decreasing but still there are certain indications for which catheterization is required:
AHA Recommendations for Diagnostic Catheterization
Class I
- It is recommended that hemodynamic and anatomic data be obtained (via angiography when necessary) at the time of a planned interventional cardiac catheterization (Level of Evidence: A).
- It is recommended that cardiac catheterization be used to assess pulmonary resistance and reversibility of pulmonary hypertension in patients with CHD or primary pulmonary hypertension when accurate assessment of pulmonary resistance is needed to make surgical and medical decisions (Level of Evidence: B).
- Cardiac catheterization is indicated in patients with complex pulmonary atresia for the detailed characterization of lung segmental pulmonary vascular supply, especially when non invasive imaging methods incompletely define pulmonary artery anatomy (Level of Evidence: B).
- Cardiac catheterization is indicated in determination of coronary circulation in pulmonary atresia with intact septum (Level of Evidence: B).
- Cardiac catheterization is indicated in patients being assessed for cardiac transplantation unless the patient's risk for catheterization outweighs the potential benefit (Level of Evidence: C). (1)
- Cardiac catheterization is recommended for surveillance of graft vasculopathy after cardiac transplantation (Level of Evidence: B).
Class IIa
- It is reasonable to perform a cardiac catheterization to determine pulmonary pressure/resistance and transpulmonary gradient in palliated single-ventricle patients before a staged Fontan procedure (Level of Evidence: B).
- Cardiac catheterization is reasonable in any CHD patient in whom complete diagnosis cannot be obtained by non invasive testing or in whom such testing yields incomplete information (Level of Evidence: C).
- Cardiac catheterization is reasonable for the assessment of cardiomyopathy or myocarditis (Level of Evidence: B).
- Cardiac catheterization is reasonable for the assessment of coronary circulation in some cases of Kawasaki disease in which coronary involvement is suspected or requires further delineation or in the assessment of suspected congenital coronary artery anomalies (Level of Evidence: B).
- Cardiac catheterization is reasonable to perform for the assessment of anatomy and hemodynamics in postoperative cardiac patients when the early postoperative course is unexpectedly complicated and noninvasive imaging techniques (eg MRA, CT angiography) fail to yield a clear explanation (Level of Evidence: C).
Risks / Complications (2)
Cardiac catheterizations are not without risk to the patient. The following is a listing of the more common complications.
- Radiation exposure
- Risk of general anesthesia (when used)
- Hypothermia (especially in small infants)
- Aggravation of hypoxia
- Arrhythmias (temporary instability or even permanent, as in heart block)
- Vascular injury/perforations/tears
- Cardiac perforation/ Cardiac valve injury
- Blood loss that requires transfusion
- Allergic reactions to contrast, drugs, or anesthetics
- Renal insufficiency caused by contrast material
- Diffuse central nervous system injury
- Stroke
- Death
Atrial septostomy / transseptal puncture (3,4)
Atrial septostomy is performed most commonly with help of balloon in cases of
1. d-TGA with intact interventricular septum to increase the mixing between two atrium in cases of low saturation. (less commonly performed now a days because of direct arterial switch operation in early new born period).
2. Decompression of left atrial hypertension in cases of mitral atresia.
3. Septostomy/ interatrial stenting in cases primary pulmonary hypertension with syncope/ failed medical management to increase cardiac output.
4. Transseptal puncture is performed in cases like pulmonary vein dilatation/stenting, left sided electrophysiologic study/ RFA in cases of arrhythmia.
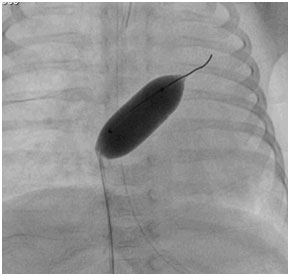
Image showing balloon atrial septostomy in case of d –TGA with intact IVS
Transcatheter device closure of septal defects (5,6,7)
Device closure of septal defects such as atrial septal defect (ASD) and muscular ventricular septal defect (VSD) is well established alternative technique to surgery in carefully selected patients and number of cases are progressively increasing because of availability of user friendly hardware and advancement in procedural skills. Device closure of perimembranous VSD is still not the procedure of choice although it is performed very frequently around the world. Device closure of other VSD’s such as inlet and outlet VSD and other forms of ASD such as ostium primum and sinus venosus ASD is not recommended
Recommendations for device closure of ASD
Class I
Transcatheter secundum ASD closure is indicated in patients with hemodynamically significant ASD with suitable anatomic features (Level of Evidence: B).
Class IIa
It is reasonable to perform transcatheter secundum ASD closure in patients with transient right-to-left shunting at the atrial level who have experienced sequelae of paradoxical emboli such as stroke or recurrent transient ischemic attack (Level of Evidence: B).
Recommendations for Device Closure of Muscular VSDs
Class IIa
It is reasonable for infants who weigh ≥5 kg, children, and adolescents with hemodynamically significant (left ventricular or left atrial volume overload or pulmonary-to-systemic blood flow ratio ≥2:1) muscular VSD to undergo percutaneous VSD device closure (Level of Evidence: B).
Risks/ complications
Device embolization, device malposition, heart block, arrhythmia, infection, bleeding, thrombosis, valve regurgitation, cardiac rupture, death.
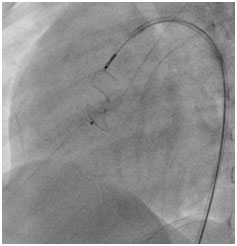
Image showing VSD device just before release
Device closure of patent ductus arteriosus (PDA)
Most of PDA are closed in cath lab by device and is considered easy and safe method of PDA closure.
Recommendations for Transcatheter PDA Occlusion
Class I
- Transcatheter PDA occlusion is indicated for the treatment of a moderate-sized or large PDA with left-to-right shunt that results in any of the following: Congestive heart failure, failure to thrive, pulmonary over circulation (with or without pulmonary hypertension), or an enlarged left atrium or left ventricle, provided the anatomy and patient size are suitable(Level of Evidence: B).
Class IIa
- Transcatheter PDA occlusion is reasonable in the presence of a small left-to-right shunt with normal-sized heart chambers when the PDA is audible by standard auscultation techniques (Level of Evidence: C).
Class IIb
- In rare instances, transcatheter PDA occlusion may be considered in the presence of a bidirectional PDA shunt due to pulmonary hypertension and obstructive pulmonary vascular disease but reversible to pure left-to-right shunting with pulmonary vasodilator therapy(Level of Evidence: C).
- Transcatheter PDA occlusion may be considered in a PDA associated with a small left-to-right shunt with normal heart size and an inaudible murmur (Level of Evidence: C).
Risks/ complications
Device embolization / malposition, aorta/ pulmonary artery occlusion, bleeding, thrombosis, death.
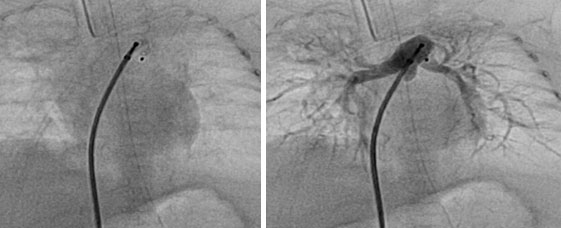
Images showing PDA device closure being done
Transcatheter balloon dilatation of cardiac valves (8,9)
Recommendations for Pulmonary Valvuloplasty
Class I
- Pulmonary valvuloplasty is indicated for a patient with critical valvar pulmonary stenosis (defined as pulmonary stenosis present at birth with cyanosis and evidence of patent ductus arteriosus dependency), valvar pulmonic stenosis, and a peak-to-peak catheter gradient or echocardiographic peak instantaneous gradient of ≥60-70 mm Hg or clinically significant pulmonary valvar obstruction in the presence of RV dysfunction (Level of Evidence: A).
Class IIa
- It is reasonable to perform pulmonary valvuloplasty on a patient with valvar pulmonic stenosis who meets the above criteria in the setting of a dysplastic pulmonary valve (Level of Evidence: C).
- It is reasonable to perform pulmonary valvuloplasty in newborns with pulmonary valve atresia and intact ventricular septum who have favorable anatomy that includes the exclusion of RV-dependent coronary circulation (Level of Evidence: C).
Class IIb
- Pulmonary valvuloplasty may be considered as a palliative procedure in a patient with complex cyanotic CHD, including some rare cases of tetralogy of Fallot (Level of Evidence: C)
Recommendations for Aortic Valvuloplasty
Class I
- Aortic valvuloplasty is indicated regardless of valve gradient in the newborn with isolated critical valvar AS who is ductal dependent or in children with isolated valvar AS who have depressed left ventricular systolic function (Level of Evidence: B).
- Aortic valvuloplasty is indicated in children with isolated valvar AS who have a resting peak systolic valve gradient (by catheter) of ≥50 mm Hg (Level of Evidence: B).
- Aortic valvuloplasty is indicated in children with isolated valvar AS who have a resting peak systolic valve gradient (by catheter) of ≥40 mm Hg if there are symptoms of angina or syncope or ischemic ST-T-wave changes on electrocardiography at rest or with exercise(Level of Evidence: C).
Class IIb
- Aortic valvuloplasty may be considered in a child or adolescent with a resting peak systolic valve gradient (by catheter) of ≥40 mm Hg but without symptoms or ST–T-wave changes if the patient desires to become pregnant or to participate in strenuous competitive sports (Level of Evidence: C).
- Aortic valvuloplasty may be considered for asymptomatic patients with a catheter-obtained peak systolic gradient of <50 mm Hg when the patient is heavily sedated or anesthetized, if a nonsedated Doppler study finds the mean valve gradient to be >50 mm Hg(Level of Evidence: C)
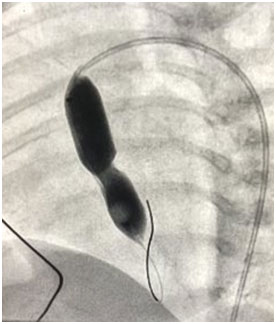
Image showing balloon aortic valvoplasty being done with Tyshak balloon
Recommendations for Mitral Valvuloplasty
Class I
- Transcatheter balloon valvuloplasty is indicated for symptomatic patients with moderate to severe isolated rheumatic mitral stenosis or asymptomatic patients with moderate to severe rheumatic mitral valve stenosis/restenosis associated with pulmonary hypertension (Level of Evidence: B).
Class IIa
- Transcatheter balloon valvuloplasty is reasonable for the treatment of symptomatic children ≥5 years of age with congenital mitral stenosis or restenosis who have mitral valve morphology that is favorable for balloon valvuloplasty (ie, thickened leaflets and fused commissure) (Level of Evidence: B).
Risks/ complications of transcatheter balloon dilatation of valves
Valve Regurgitation, cardiac rupture, pericardial effusion/tamponade, heart failure, arrhythmia, infection, bleeding, thrombosis, death
Coarctation of Aorta (10)
Recommendations for Transcatheter Balloon Angioplasty of Coarctation/Recoarctation of the Aorta
Class I
- Balloon angioplasty of recoarctation is indicated when associated with a transcatheter systolic coarctation gradient of >20 mm Hg and suitable anatomy, irrespective of patient age (Level of Evidence: C).
- Balloon angioplasty of recoarctation is indicated when associated with a transcatheter systolic coarctation gradient of <20 mm Hg and in the presence of significant collateral vessels and suitable angiographic anatomy, irrespective of patient age, as well as in patients with univentricular heart or with significant ventricular dysfunction (Level of Evidence: C).
Class IIa
- It is reasonable to consider balloon angioplasty of native coarctation as a palliative measure to stabilize a patient irrespective of age when extenuating circumstances are present such as severely depressed ventricular function, severe mitral regurgitation, low cardiac output, or systemic disease affected by the cardiac condition (Level of Evidence: C).
Recommendations for Stent Placement in Native Coarctation and Recoarctation of the Aorta
Class I
- Stent placement is indicated in patients with recurrent coarctation who are of sufficient size for safe stent placement, in whom the stent can be expanded to an adult size, and who have a transcatheter systolic coarctation gradient >20 mm Hg (Level of Evidence: B).
Class IIa
It is reasonable to consider placement of a stent that can be expanded to an adult size for the initial treatment of native or recurrent coarctation of the aorta in patients with:
- a transcatheter systolic coarctation gradient of >20 mm Hg (Level of Evidence: B).
- a transcatheter systolic coarctation gradient of <20 mm Hg but with systemic hypertension associated with an anatomic narrowing that explains the hypertension (Level of Evidence: C).
- a long-segment coarctation with a transcatheter systolic coarctation gradient >20 mm Hg (Level of Evidence: B).
- Stent implantation for the treatment of coarctation (native or recurrent) is reasonable in patients in whom balloon angioplasty has failed, as long as a stent that can be expanded to an adult size can be implanted(Level of Evidence: B).
Risks/ complications
Aortic rupture/ dissection/ aneurysm, restenosis, bleeding, infection, thrombosis, embolism, death
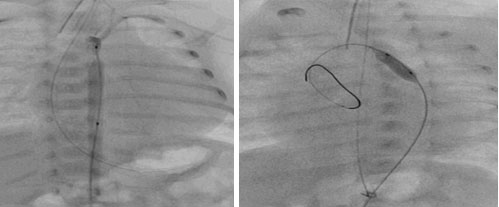
Images showing ballooning of coarctation in 1200 gram preterm newborn.
There is still a long list of procedures performed in cath lab which is mentioned as under:
- PDA stenting in duct dependent circulation
- Device closure of coronary cameral fistula, pulmonary AV malformations
- Closure of MAPCA, veno venous collaterals
- Ballooning/ stenting of branch Pulmonary artery
- Perforation of pulmonary valve
- Percutaneous pulmonary valve / aortic valve implantation
- Device closure of aorto pulmonary window
- Stenting/ ballooning of surgical shunts
- Device closure of paravalvar leaks
- Device closure of Fontan fenestration/ creation of Fontan fenestration
Conclusion
Catheter intervention has a very important role in management of congenital heart diseases, not only for diagnostic purposes but also for the treatment of CHD. Without catheter interventions, management of CHD is incomplete.
References
- Pahl E, Naftel DC, Kuhn MA, Shaddy RE, Morrow WR, Canter CE, Kirklin J; Pediatric Heart Transplant Study. The impact and outcome of transplant coronary artery disease in a pediatric population: a 9-year multi-institutional study. J Heart Lung Transplant. 2005; 24:645–651.
- Mehta R, Lee KJ, Chaturvedi R, Benson L. Complications of pediatric cardiac catheterization: a review in the current era. Catheter Cardiovasc Interv. 2008; 72:278–285.
- El-Said HG, Ing FF, Grifka RG, Nihill MR, Morris C, Getty-Houswright D, Mullins CE. 18-Year experience with transseptal procedures through baffles, conduits, and other intra-atrial patches. Catheter Cardiovasc Interv. 2000; 50:434–439.
- Mullins CE. Transseptal left heart catheterization. In: , Mullins C, ed. Cardiac Catheterization in Congenital Heart Disease: Pediatric and Adult. Malden, MA: Blackwell Futura; 2006:223–254.
- Du ZD, Hijazi ZM, Kleinman CS, Silverman NH, Larntz K; Amplatzer Investigators. Comparison between transcatheter and surgical closure of secundum atrial septal defect in children and adults: results of a multicenter nonrandomized trial. J Am Coll Cardiol. 2002; 39:1836–1844.
- Kaya MG, Baykan A, Dogan A, Inanc T, Gunebakmaz O, Dogdu O, Uzum K, Eryol NK, Narin N. Intermediate-term effects of transcatheter secundum atrial septal defect closure on cardiac remodeling in children and adults. Pediatr Cardiol. 2010; 31:474–482
- Holzer R, Balzer D, Cao QL, Lock K, Hijazi ZM; Amplatzer Muscular Ventricular Septal Defect Investigators. Device closure of muscular ventricular septal defects using the Amplatzer muscular ventricular septal defect occluder: immediate and mid-term results of a U.S. registry. J Am Coll Cardiol. 2004; 43:1257–1263
- Rey C, Marache P, Francart C, Dupuis C. Percutaneous transluminal balloon valvuloplasty of congenital pulmonary valve stenosis, with a special report on infants and neonates. J Am Coll Cardiol. 1988; 11:815–820.
- Fischer DR, Ettedgui JA, Park SC, Siewers RD, del Nido PJ.Carotid artery approach for balloon dilation of aortic valve stenosis in the neonate: a preliminary report. J Am Coll Cardiol. 1990; 15:1633–1636
- Rothman A, Galindo A, Evans WN, Collazos JC, Restrepo H.Effectiveness and safety of balloon dilation of native aortic coarctation in premature neonates weighing Am J Cardiol. 2010; 105:1176–1180.





